Author: Elvirosa Brancaccio – Serintel Srl – Rome (Italy)
1. Introduction
Gas-to-liquids (GTL) is a technology that enables the production of clean-burning diesel fuel, liquid petroleum gas, base oil and naphtha from natural gas. The GTL process transforms natural gas into very clean diesel fuel because products are colorless and odorless hydrocarbons with very low level of impurities.
Much of the world’s natural gas is classified as “stranded,” meaning it is located in a remote area, far from existing pipeline infrastructure. The volumes often are too small to make constructing a large-scale treatment gas plant cost-effective. As a result, the gas is typically re-injected into the reservoir, left in the ground, or flared, which is harmful to the environment. However, the availability of this low cost, stranded gas has incentivized companies to develop innovative technologies that can economically and efficiently utilize this gas converting it into a transportation fuel like diesel and jet fuel.
Refineries can also use GTL to convert some of their gaseous hydrocarbon waste products into valuable fuel oil which can be used to generate income.
Small-scale GTL plants are containerized units comprised of a reformer for synthesis gas production, a Fischer Tropsch (FT) reactor for syncrude production, and, in some cases, an upgrading package, which is used to further refine the FT products into the desired transportable fuel. Since these containerized units already have about 70 percent of their construction complete before reaching the plant site, on-site construction costs are significantly reduced. In cases where capacity needs to be increased, additional units can be easily shipped via truck or ship and connected in parallel to the existing process. Depending on the technology, capacity can range anywhere from 100 barrels per day (bpd) to 15,000 bpd.
2. GTL Process Phases
Fischer-Tropsch is the process of chemical converting natural gas into liquids (GTL), coal to liquids (CTL), biomass to liquids (BTL) or bitumen from oil sands to liquids (OTL).
All four processes consist of three technological separate sections.
- The production of synthesis gas (syngas).
The carbon and hydrogen are initially divided from the methane molecule and reconfigured by steam reforming and/or partial oxidation. The syngas produced, consists primarily of carbon monoxide and hydrogen.
- Catalytic (F-T) synthesis.
The syngas is processed in Fischer-Tropsch (F-T) reactors of various designs depending on the technology creating a wide range of paraffinic hydrocarbons product (synthetic crude, or syncrude), particularly those with long chain molecules (e.g. those with as many as 100 carbons in the molecule).
- Cracking – product workup.
The syncrude is refined using conventional refinery cracking processes to produce diesel, naphtha and lube oils for commercial markets. By starting with very long chain molecules the cracking processes can be adjusted to an extent in order to produce more of the products in demand by the market at any given time. In most applications it is the middle distillate diesel fuels and jet fuels that represent the highest-value bulk products with lubricants offering high-margin products for more limited volume markets. In modern plants, F-T GTL unit designs and operations tend to be modulated to achieve desired product distribution and a range of product slates.

Fig. 1 – GTL technological process with Fischer-Tropsch synthesis reactor
Research and development in GTL process and plant involves several part of the plant:
- the production efficiency increment for each single unit used upstream and downstream
- the catalyst into the FT reactor in order to increase its selectivity and durability
- the design of the reactors to reduce the entire plant or module foot print
3. Start and Development
Synthetic fuel production technology, known as GTL, was invented in the 1920s. One of the best-known ways to create synthetic fuel is through Fischer-Tropsch (FT) synthesis. FT technology was initially developed in Germany to solve petroleum shortages leading up to World War. By 1944, Germany was producing 124 Mbpd of synthetic fuels from coal at 25 FT plants.
Next-generation technology was developed in South Africa, which sought to support its economy without oil. In the 1970s, the technology evolved in Western Europe and the US with big plant and large scale production.
Starting from the last decades, advances in GTL technologies have enabled small-scale GTL, and even micro-scale GTL, to be operationally and potentially economically feasible.
Several factors are converging to drive the growth in the GTL industry:
- Desire to monetize existing stranded gas reserves;
- Energy companies keen to gain access to new gas resources;
- Market demand for cleaner fuels and new cheaper chemical feedstocks;
- Rapid technology development by existing and new players;
- Increased interest from gas rich host governments
As petroleum prices remain high, new discoveries make natural gas abundant and cheap by comparison, and more advanced energy companies are exploring ways to reduce the CAPEX of synthetic fuel production. As part of this goal, companies are looking into building smaller-scale, modular plants that can operate in remote locations[1].
Several Gas-to-Liquids (GTL) technologies have emerged over the past three decades as a credible alternative for gas monetisation for gas-producing countries to expand and diversify into the transportation fuel markets. The final GTL product may be syncrude, which can be injected into an oil pipeline, thereby avoiding the need to transport another product to market, or higher-value liquid fuels or chemical feedstocks such as gasoline, diesel (without sulphur and with a high cetane number), naphtha, jet fuel, methanol or di-methyl ether (DME).
4. Plants and Projects
WORLD COMMERCIAL-SCALE GTL PLANTS
At present, five commercial-scale GTL plants are in operation (Fig. 1). These five plants include:
- Bintulu GTL, Malaysia
- Escravos GTL, Nigeria
- Mossel Bay GTL, South Africa
- Oryx GTL, Qatar
- Pearl GTL, Qatar.
These five plants represent nearly 259 Mbpd of capacity. At 140 Mbpd, Shell’s Pearl GTL complex represents more than 50% of the world’s total commercial-scale GTL capacity.

Fig. 2 – Commercial-scale GTL plants in operation around the world [2]
The first GTL plant was developed by PetroSA in 1992. This 36-Mbpd plant is in Mossel Bay, South Africa. The plant utilizes FT technology to process methane-rich natural gas into high-quality, low-sulfur synthetic fuels. Products include unleaded petrol, kerosene, diesel, propane, distillates, process oil and alcohols.
Shell commissioned its first commercial GTL plant in Bintulu, Malaysia in 1993. The plant’s initial construction cost was $850 MM. The 12.5-Mbpd plant underwent a $50-MM debottlenecking that increased total capacity to 14.7 Mbpd. Since 1993 has produced the following products: liquefied petroleum gas (up to 5%), naphtha (up to 30%), diesel fraction (up to 60%) and paraffin (up to 5-10%).

Fig. 3 – Bintulu GTL plant [3]
The Pearl GTL complex is the largest GTL facility in the world. The 140-Mbpd facility is located in Ras Laffan Industrial City, Qatar. The $19-B natural gas processing and GTL integrated complex was developed by a JV of Shell and Qatar Petroleum.
Oryx GTL was the Middle East’s first GTL plant. Developed by Qatar Petroleum and Sasol, the $6-B plant also processes natural gas from Qatar’s North Field. Construction of the facility began in late 2003, and it started production in early 2007. The facility processes 330 Mcfd of methane-rich gas from Qatar’s North field and produces 34 Mbpd of liquids, with the majority being low-sulfur, high-octane GTL diesel.
The latest commercial-scale GTL plant to commence operations is the Escravos GTL plant. The $10-B facility was developed by a JV consisting of Chevron, Sasol and Nigerian National Petroleum Corp. The plant utilizes technology from both JV partners to convert up to 325 MMcfd of natural gas into 33 Mbpd of GTL diesel and GTL naphtha. The plant has been operational since 2014.
NEW GTL FACILITIES UNDER DEVELOPMENT
The ENVIA Energy’s GTL plant on the Waste Management landfill in Oklahoma came on line in 2017. The plant, partially fed with landfill gas, announced its first finished, sale able products on June 30 2017, but at January 2018, has not yet reached the 250 bpd design capacity.
The start-up of other 4 plants (Greyrock 1, Juniper GTL, Primus 1 and Primus 2) will happen in 2018. The new owner of Juniper GTL, York Capital, will likely target future plant sizes of more than 5000 bpd (consuming 50 MMscfd of gas). Greyrock and Primus GE announced to continue strong business development efforts in the gas flare arena.
Haldor Topsoe has joined forces with Modular Plant Solutions (MPS) and has designed and engineered a small-scale methanol plant (215 tpd) called “Methanol-To-GoTM”. The size of the plant is similar to the Primus 1 and 2 plants with a gas feed rate of 7 MMscfd.
BgtL is a new player in the micro-GTL arena (20-200 bpd). However, their patented technologies are based on 2 decades of R&D work in research institutes. Their portfolio of products includes plant modules that convert gas volumes as small as 2 Mscfd into a range of products including oil, diesel, methanol and others.
Summarizing, the current leading GTL technology providers with commercial offers are:
Micro-GTL: Unattended operation units below ~1MMscfd and below ~US$ 10mln
Mini-GTL: Small modular plants with some operators and a cost >US$ 10mln
- Greyrock
- EFT/Black and Veatch
- INFRA
- Primus GE
- Topsoe/MPS
- Expander Energy
More information on these companies and their projects can be found into the most recent bulletin on GTL technology [4].
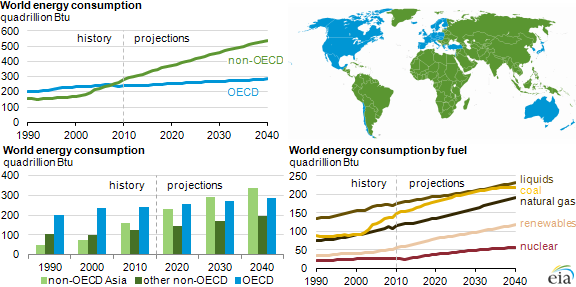
In the following figure is reported the forecast furnished by EIA for GTL production in the next few years:

Fig. 4 – Global gas to liquid plant production, 2017 [5]
4.1 Available Technologies Overview
The GTL market is pushing toward small-scale and modular units. These types of plants can be built at greatly reduced capital cost, which can run into the billions of dollars for large-scale facilities.
Gas units, technologies used, size and other functional data for several companies involved in the GTL technology are summarized in the tables below[6]:
Calvert Energy Group/OXEON

Fig. 5 – Calvert Energy Group GTL plant
The Calvert Energy Group offers modular GTL (Flare & Stranded Gas to Diesel plants ranging in size from 0.2 MMscf/d to 100 MMscf/d. The OEXON technology used is exclusively licensed to Calvert Energy Group by OXEON.

Tab. 1 – Calvert Energy Group data
CompactGTL

Fig. 6 – Compact GTL’s modular plant
CompactGTL’s modular unit offers a small-scale gas-to-liquid (GTL) solution for small- and medium-sized oil field assets where no viable gas monetization option exists so that the associated gas is either flared or reinjected.

Tab. 2 – Compact GTL’s modular unit data
GasTechno Energy & Fuels (GEF)

Fig. 7 – Gas Technologies LLC module
Gas Technologies LLC manufactures, installs and operates modular gas-to-liquids plants that utilize the patented GasTechno® single-step GTL conversion process. GasTechno® Mini-GTL® plants convert associated flare gas and stranded natural gas into high-value fuels and chemicals including methanol, ethanol and gasoline/diesel oxygenated fuel blends while serving to reduce greenhouse gas emissions. The unit capital cost of the plants is approximately 70% lower than traditional methanol production facilities and they require relatively limited operation & maintenance costs.

Tab. 3 – Gas Technologies LLC data
Greyrock

Fig. 8 – Greyrock Energy module P-5000
Greyrock Energy was founded in 2006 and is headquartered in Sacramento, California, with offices and a demonstration plant in Toledo, Ohio. Its sole focus is small-scale GTL Fischer-Tropsch plants for Distributed Fuel Production®, and it has a commercial offer of both a fully integrated 2000 bpd plant consuming about 20 MMscfd and smaller “MicroGTL” plants (5 – 50 bpd).

Tab. 4 – Greyrock Energy data
Velocys

Fig. 9 – Velocys plant
Velocys is a smaller-scale GTL company that provides a bridge connecting stranded and low-value feedstocks, such as associated gas and landfill gas, with markets for premium products, such as renewable diesel, jet fuel and waxes. The company was formed in 2001, a spin-out of Battelle, an independent science and technology organization. In 2008, it merged with Oxford Catalysts, a product of the University of Oxford. Velocys aims to deliver economically compelling conversion solutions. It is traded on the London Stock Exchange, with offices in Houston, Texas; Columbus, Ohio; and Oxford, UK.

Tab. 5 – Velocys data
Primus Green Energy

Fig. 10 – Primus System
Primus Green Energy is based in Hillsborough, New Jersey, USA. The company is backed by Kenon Holdings, a NYSE-listed company with offices in the United Kingdom and Singapore that operates dynamic, primarily growth-oriented, businesses. Primus Green Energy™ has developed Gas-to-Liquids technology that produces high-value liquids such as gasoline, diluents and methanol directly from natural gas or other carbon-rich feed gas.

Tab. 6 – Primus Green Energy data
5. Developments Remarks
DOWNSIZING ADVANTAGES
By taking advantage of new technologies, such as microchannel reactors, to shrink the FT and SMR hardware, GTL plants can be scaled down to provide a cost-effective way to take advantage of smaller gas resources. GTL plants based on the use of microchannel FT reactors can be operated on a distributed basis, with smaller plants located near gas resources and potential markets.
Smaller, modular GTL plants are suitable for use in remote locations. In contrast to conventional GTL plants, they are designed for the economical processing of smaller amounts of gas ranging from 100 million cubic meters (MMcm) to 1,500 MMcm, and they can produce 1,000 bpd–15,000 bpd of liquid fuels. The plants can be scaled to match the size of the resource, expanded as necessary, and potentially integrated with existing facilities on refinery sites.
Smaller-scale GTL operations also pose a lower risk to producers. Since the plants are smaller, construction costs are reduced; and, since the plants are modular, investment can be phased. The construction time is short, at 18–24 months. In addition, because the modules and reactors are designed only once and then manufactured many times, much of the plant can be standardized and shop-fabricated in skid-mounted modules. This reduces the cost and risk associated with building plants in remote locations. In addition, the components can be designed to use standard, off-the-shelf equipment, so there is less strain on supply chains, and the need for onsite construction work is reduced.
Since the FT process also lies at the heart of the biomass-to-liquids (BTL) processes, the same technology can be used to produce high-quality, ultra-clean diesel and jet fuel from waste biomass, including municipal waste. Smaller-scale GTL plants offer advantages at all stages of production: upstream, midstream and downstream [7].
6. GTL-FT Technology New Concepts
The small-scale processing of natural gas needs principally new technologies for converting hydrocarbons into liquid chemicals and fuels. There are several possibilities.
The first one is to develop more effective, less complex methods for converting hydrocarbon gases into syngas.
- A very promising way to increase the efficiency and flexibility of the conversion of hydrocarbon gases into syngas is the gas-phase combustion of very rich hydrocarbon-air or hydrocarbon–oxygen mixtures in volumetric permeable matrixes. The partial oxidation of hydrocarbon gases is very attractive method for small-scale syngas production since it is an exothermic process, which therefore requires no external heating and, consequently bulky and expensive heat-exchange equipment. This circumstance makes it possible to significantly decrease the size and, hence, the cost of the reformer.
The second is to work out principally different methods for the conversion of natural gas into chemicals without the intermediate stage of syngas production, working on the composition of the used catalysts or either by developing new ones.
- An alternative possibility to produce useful chemicals and liquid fuels from natural gas is their direct oxidation. Several direct methods of natural gas conversion into useful chemicals without intermediate production of syngas can be discussed. Among them, the most known and developed are Direct oxidation of Actually, direct partial oxidation with subsequent carbonylation and/or oligomerization of oxidation products can beconsidered as an alternative route for Gas-To-Liquids processes, which enables to avoid syngas production, the most costly andenergy-consuming stage of traditional GTL [8].
With smaller-scale GTL plants, the greatest challenge is to find ways to combine and scale down the size and cost of the reaction hardware while still maintaining sufficient capacity. This, in turn, depends on finding ways to reduce reactor size by enhancing heat-transfer and mass-transfer properties to increase productivity and intensify the syngas-generation and FT processes. The use of microchannel reactors offers a way to achieve these goals.
- Microchannel technology is a developing field of chemical processing that intensifies chemical reactions by reducing the dimensions of the channels in reactor systems. Since heat transfer is inversely related to the size of the channels, reducing the channel diameter is an effective way of increasing heat transfer, thereby intensifying the process and enabling reactions to occur at significantly faster rates than those seen in conventional reactors.
The technology can be applied to both highly exothermic processes such as FT, and highly endothermic processes such as SMR. Microchannel FT reactors contain thousands of thin process channels filled with FT catalyst, interleaved with water-filled coolant channels. Since the small-diameter channels dissipate heat more quickly than do conventional reactors, more active FT catalysts can be used to significantly accelerate FT reactions, thereby boosting productivity.
In microchannel SMR reactors, the heat-generating combustion and SMR processes take place in adjacent channels. The high heat-transfer properties of the microchannels make the process very efficient (Fig. 4).

Fig. 11 – An FT microchannel reactor diagram (left), and the reactor in a full-pressure shell (right)[9]
Additional improvement can be obtained by catalyst research.
- INFRA Technology represents the new generation of GTL technology allowing the production of light synthetic crude oil straight out of the FT reactor, with four-fold performance and without byproducts (Fig. 12). The process does not require additional processing of waxes, and synthetic crude oil is fully compatible with the existing oil infrastructure.

Fig. 12 New Technology applications[10]
The technology was made possible by creating a novel catalyst using cobalt as active metal in a multicomponent composite. Elimination of certain processing stages and production of high-quality, single-liquid product makes INFRA’s GTL solutions economically feasible from small-scale, pre-engineered, standardized, modular (as small as containers), easily deployed and transportable units all the way to large-scale, integrated gas processing plants.
7. Cost Analysis
By offering the ability to target supply into global-liquid-fuel-transportation markets GTL plants significantly diversify market opportunity and help to smooth financial returns in volatile conditions where gas markets prices and oil and petroleum product market prices become decoupled.
7.1 Cash Flow Analysis Methodology to Evaluate the Commerciality of GTL Projects
There are several factors that determine the cash flow and income streams associated with GTL plants. The key factors required for a methodology that analyses the commercial attractiveness of a GTL plant in a multi-year cash flow model include:
- Cost of feedstock (natural gas, coal, petroleum coke or biomass)
- Prices of the petroleum products and chemicals produced and sold from the plants.
Those product prices are in most cases strongly influenced by benchmark crude oil prices. GTL products generally trade in price ranges that reflect prevailing refinery and petrochemical plant crack spreads. Sometimes GTL products trade at small premiums to refinery derived products because of their superior quality (i.e. low sulphur, low aromatics in the case of diesel and gasoline).
Aspects to be considered are:
- If the GTL project is an integrated project then revenue from natural gas liquids extracted from the feed gas stream need to be included in the project cash flow and income calculations
- Capital costs to construct the GTL plant, which can be usefully compared by the unit US$/ barrel/day of plant product throughput capacity
- How capital costs are offset, recovered and/or depreciated over time and deducted as part of a taxable income methodology
- GTL plant efficiency (i.e. unit quantities of feedstock required to produce one unit of product) on an energy and/or mass basis
- GTL plant annual utilization rate (days/year) based upon maintenance and turnaround requirements
- GTL plant operating and maintenance costs including the costs of catalysts, chemicals, utilities
- Cost of transportation (shipping) between the GTL plant and the market in which the products are sold
- Fiscal deductions applied which vary significantly from jurisdiction to jurisdiction
7.2 Cost Forecast
FT technology typically has four components: synthesis gas (syngas) generation, gas purification, FT synthesis and product upgrading. The third stage constitutes a distinctive technology that provided the basis for future technological developments and innovations. The remaining three technologies were well-known before FT invention, and have been developed separately.
The syngas is normally produced via high-temperature gasification in the presence of oxygen and steam.
For the components of the plant, some aspects can be considered for cost analysis:
- The air separation unit typically represents a considerable CAPEX investment.
- The economic advantages or breakthrough is in small scale GTL plants have occurred with the advances in 4 areas:
- Commercial introduction of micro-channel F-T technology;
- Higher reactive cobalt catalysts;
- Mass production of F-T reactors;
- Modular construction of the plants.
- Another fundamental challenge is that, due to environmental regulations, heavy feed slates (primarily asphalts and heavy fuel oils) are increasingly difficult to market and, therefore, become unwanted residues rather than revenue generators. GTL technology has a clear advantage here due to its complete lack of heavy slates. This may become a strong argument for GTL in the future, especially for FT installations within existing refineries that can be used to increase the share of light and middle distillates in the overall product portfolio[11].
8. Environmental Aspects and Benefits
GTL technologies can transform off gas streams, which would otherwise be flared into valuable liquid transportation fuels and chemicals, including high-quality gasoline or methanol or a separate stream of hydrogen-rich vent gas that can be used as an additional onsite hydrogen or fuel source, so this is an ideal solution for reducing gas flaring while boosting returns.
In addition, greenhouse gas emissions can be further reduced with GTL systems through the input of CO2 streams as co-feed which is converted into gasoline or methanol, representing a valuable use for what is typically considered a low-value or even negative-value gas stream.
Properties of GTL Fuel include the enhanced aquatic and soil biodegradability, lower aquatic and soil ecotoxicity. Fuels produced from the FT process offer significantly better performance than their petroleum-based equivalents. FT-derived diesel does not contain aromatics or sulfur, and it burns cleaner than petroleum-derived fuels, resulting in lower emissions of nitrogen oxide (NOx), sulfur oxide (SOx) and particulates. Exhaust emissions experiments on GTL products revealed an overall significant reduction of CO (22%–25%), hydrocarbons (30%–40%) and NOx (6% to 8%). GTL diesel has the potential to be sold as a premium blendstock[12].
The combination of these features indicate that GTL Fuel is less likely to cause adverse environmental impacts than clean conventional fuels. In addition, FT diesel can be blended with lower-cetane, lower-quality diesels to achieve commercial diesel environmental specifications.
When the feedstock includes a renewable component, whether renewable biogas (as in the case of the ENVIA Energy project), or forestry and sawmill waste (as in the case of Red Rock Biofuels’ proposed project in Oregon), the fuels produced deliver a significant reduction in lifecycle greenhouse gas (GHG) emissions over conventionally produced fuels.
Click here for some video:
or contact us for more information about GTL technology.
[3] www.theoildrum.com
[6] GGFR Technology Overview – Utilization of Small-Scale Associated February 2018


 Fig. 1 – Rashig rings
Fig. 1 – Rashig rings