Biodiesel from Microalgae
Author: Vincenzo Piemonte, Associate Professor, University UCBM – Rome (Italy)
1. Theme description
The actual dependence on fossil fuels (more than 80% of the actual world energy demand is covered by fossil fuels) must be cut before 2030, the upcoming future scenario being unknown and depending on the occurrence of the oil peak is probably in the next few years.
Biofuels are an interesting options to fossil fuels for transportation purposes; nonetheless, they pose different concerns: intensive bioenergy crops, endangering local biodiversity, food vs. fuel issue.
The sustainability of biofuels must exclude bioenergetical crops over arable lands and edible feedstocks, but promotes the use of biological wastes and crops from non-arable lands (second and third generation biofuels), remaining economically competitive.
At the moment, biodiesel is the commonest and most promising biofuel already introduced into the automotive fuels marked (in mixture with petrodiesel) at the cost of small technological adaptation of automotive engines. The biodiesel use strongly reduces the GHG (up to 78%), unburned hydrocarbons and residual powder emissions.
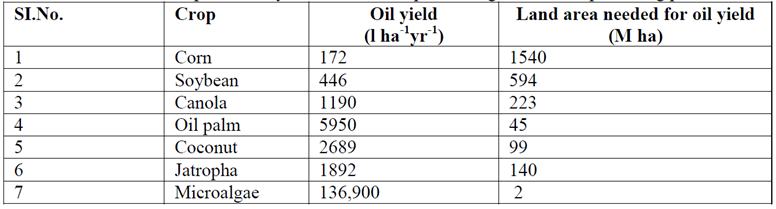
Feedstocks, class of lands for bioenergy crops (whether or not arable) and the production process affect the biodiesel production sustainability. The use of microalgae as feedstocks complies with the sustainability requirements (see Table 1).
Microalgae are Eukariotes, strongly adaptable to any ecosystem on Earth: they convert, through photosynthesis, carbon dioxide into biofuelswith the aid of light by means of photosynthetic metabolism, resulting into CO2 mitigation as well (around 1.7 kg of CO2 caught by 1 kg of dry biomass).
Microalgae photo bioreactors occupy water surfaces or non-arable land, not interfering with the edible feedstocks production. They are elective microorganisms for biodiesel production due to their high oil content (they produce more than 10,000 gallons of oil per acre) [1].
However, different economical, environmental and technological issues hinders the full development of the industrial potential of biodiesel. On the technological point of view, it is necessary to cope with two issues: to raise the oil productivity per acre of microalgae, in by means of synthetic biology and of improved the light supply systems; improve the competitivess and reduce the environmental impact of downstream processing (energy duty). Simpler and more efficient oil extraction techniques would promote the whole technology to industrial large scale applications.
Among different technologies for oil extraction and separation from biomass [2], methods with ionic liquids are highly promising thanks to their extraction efficiency and the low environmental impact of solvents [3].
Table 1 – Oil productivity and land area required for growth of oil producing plants
2. Production Process
2.1 Cultivation Systems
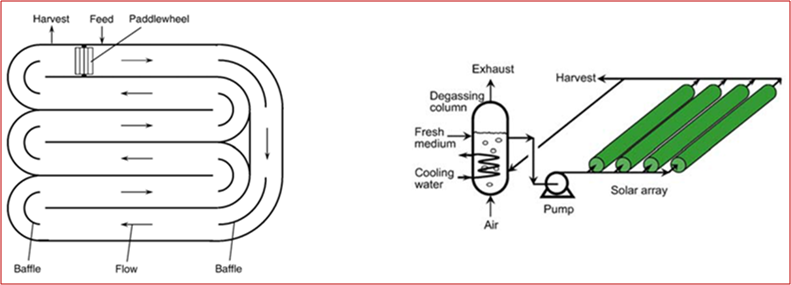
The most used plants for microalgae cultivation are open or closed systems. The first one are common called Raceway Ponds (RP), while the second one are Photobioreactors (PBR) (see Figure 1) [4]. The main differences between the two cultivation systems can be summarized as follow:
- In the PBR the microalgae concentration (g/l) is 30 times higher than in the RP;
- The land required to install a PBR is 2/3 of the land needed from RP;
- The volumetric productivity (g/l*d) of a PBR is 10 times higher than that of a RP;
- The installation costs of a RP is about 10 timer lower than that of PBR;
- The operative costs of a PBR are quite lower than that of RP due to an easier biomass harvesting process.
The main steps for microalgae production comprise the microalgae harvesting, the microalgae dewatering and the oil extraction, by wet or dry route [5]. Among the different techniques used to extract the bio-oil from microalgae, one of the most promising is the use of ionic liquids (IL). Ionic liquids are organic salts in the liquid phase, whose melting point is not higher than room temperature: they are made up of an organic cation and an inorganic/organic anion [6], so they are amphoters. Due to their chemical nature, upon contact with cells, they break their walls and membranes, resulting into oil release.
Figure 1 – a) Arial view of raceway pond; b) Tubular photobioreactor
As example, the production of bio-oil from microalgae by using IL is detailed in the following:
- Microalgae harvesting: microalgae grow in PBR or RP with additional CO2. In the first harvesting step, a lamella settler thickens the algae solution so to achieve 2% dry weight after bioflocculation.
- Microalgae dewatering: after the bioflocculation, the microalgae sludge is treated by mechanical (centrifuge) dewatering to reach the DW threshold(20%) required for downstream processing. The water is further recycled into the microalgae harvesting system.
- Oil extraction (wet route): the lysis reactor works generally at 80°C and atmospheric pressure, treating as the feed stream the microalgae slurry from the mechanical dehydration step, mixed with the ionic liquid. A filter removes the cell debris (recycled to an anaerobic digester or sold as animal feed or lignocellulosic biomass feedstock), while a two-phase decanter separate the oil (on the top) and the aqueous solution (on the bottom).
- Ionic liquid recovery: the ionic liquid recovery occurs by evaporation of water (much more volatile than the IL). The mixture is preheated to its bubble point before the flash stage: the liquid phase (rich in ionic liquid) preheats the feed and then is recirculated to the lysis reactor, whereas the vapour phase (mainly water) is condensed and recirculated to the microalgae harvesting section. Other intracellular compounds (proteins, carbohydrates, etc.) can be recovered in the downstream stage.
2.2 Biodiesel Production
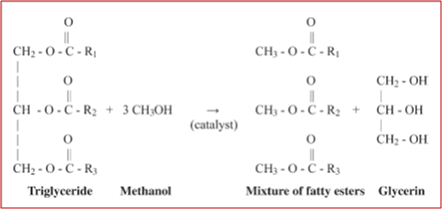
The bio-oil exstraced from microalgae is sent to a plant for biodiesel production mainly based on the transesterification process (Figure 2). The triglycerides contained in the vegetable oils are chemically transformed in a mixture of fatty acid alkyl esters (FAMEs), in the presence of an alcohol of lower molecular weight and either a homogeneous or heterogeneous catalyst.
Figure 2 – Transesterification reaction for biodiesel production
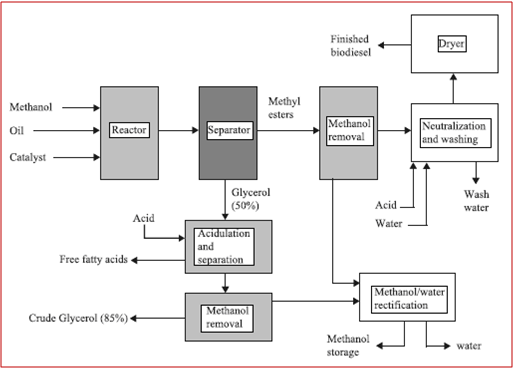
The reaction involves two main stages: specifically, in the system outlined in Figure 3, approximately 80% of the alcohol and catalyst is added to the oil in a first stage CSTR. The glycerol is then removed from the outlet stream, which is then further processed into a second CSTR, where the remaining 20% of the alcohol and catalyst is added. This configuration allows a relatively simple production process to yield high-quality fuel [7].
Figure 3 – Process flow scheme for biodiesel production
2.3 Applications
Biodiesel from microalgae as the potential to substitute diesel from fossil fuels for several purposes such as vehicular, railway and aircraft usage. Biodiesel can also be used as a heating fuel in domestic and commercial boilers or electric generators. To this end, several companies in the energy field, as example ENI, are starting the bio-oil production from microalgae on industrial scale (see figure 4), although at the time the final cost of the biodiesel produced by microalgae is still very far from being competitive with that of conventional diesel. Also for these reasons, ENI has developed an innovative technology for the production of a biofuel alternative to biodiesel, called Green Diesel, which appears to be extremely promising [8].
Figure 4 – The ENI’s Raceway Pond in Gela Refinery